Blog
Why Radiopacity Matters in Next-gen Medical Devices
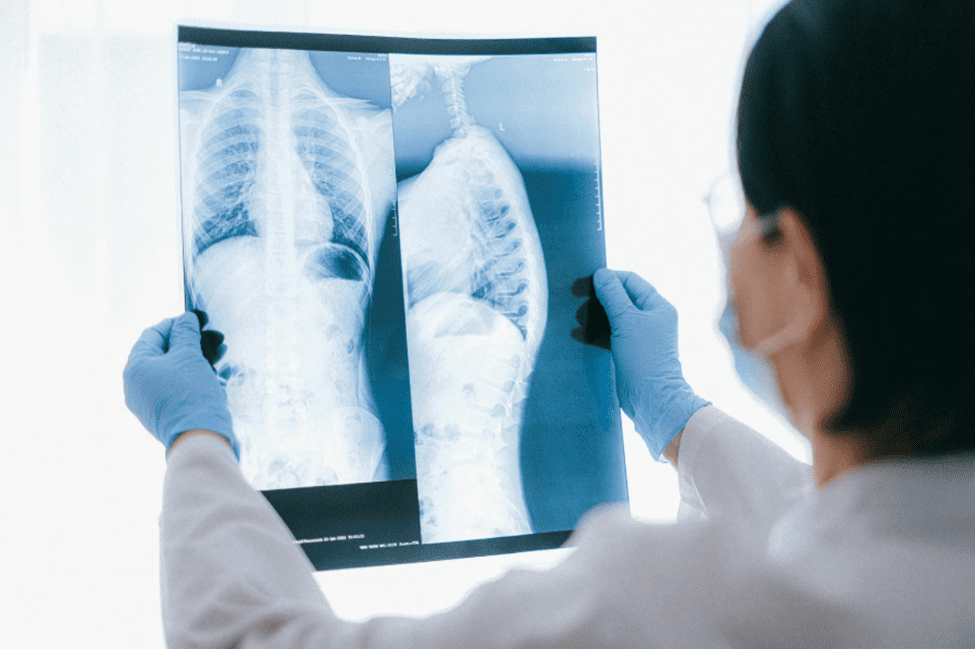
When it comes to minimally invasive procedures, being able to see the device matters. Whether a doctor is guiding a catheter or placing an implant, they need to rely on imaging to know the exact location of the device. That’s where radiopacity comes in. At Medical Murray, we develop and manufacture advanced radiopaque catheters and…
Read MoreWhat It Takes to Manufacture Pediatric Catheters
Medical Murray | Complex Catheter and Implant Solutions Manufacturing pediatric catheters isn’t just about shrinking things down. It’s about rethinking the design, materials, and entire manufacturing process to work safely and effectively in smaller, more sensitive anatomy. At Medical Murray, we’ve worked on a wide range of pediatric medical devices, from neonatal vascular access to…
Read MorePractical Advice for Medical Device Startups
A polished slide deck can open doors and land seed money. But Series A investors, and eventually clinicians and patients, pay for working products. This means devices that can be built, sterilized, shipped, and cleared by regulators. Use this reality-check to move from seed funding to first commercial sales without running out of runway. A Pretty…
Read MoreLaser Welding in Medical Device Manufacturing
Laser welding uses a hair-thin, high-intensity light beam to join materials with micron-scale precision. By confining all the heat to a microscopic interaction zone, known as the heat-affected zone (HAZ), the process briefly melts the workpiece before it solidifies almost instantly. This tight thermal footprint (often under 50 µm) keeps nearby plastics, sensors, and coatings…
Read MoreAvoiding Common Pitfalls in Medical Device Testing
How to De-risk Your Path to Regulatory Approval and Market Success When it comes to medical devices, testing isn’t just a checkbox. It’s a critical step that determines safety, performance, and your path to regulatory approval and commercialization. At Medical Murray, we’ve seen firsthand how missteps in testing can cause major delays, regulatory pushback, and…
Read MoreHow Medical Murray Partners on Medical Device Iteration Post-Launch
Launching a medical device is just the beginning. At Medical Murray, we understand that once a product is in the hands of clinicians, new ideas for improvement often follow. That’s why we partner closely with our customers after launch to support design refinements, address real-world feedback, and enable smooth, compliant updates to commercialized devices. Why…
Read MoreWhy More Medtech Companies Are Moving Manufacturing to the U.S.
If the U.S. is a big part of your growth plan, where you make your device matters. Whether you’re developing robotic-assisted catheters, single-use scopes, or ablation catheters like pulse field ablation, many medtech companies are realizing it’s smarter and safer to manufacture closer to their customers. Tariffs, shipping delays, and regulatory headaches are just a…
Read MoreRobot-Assisted Surgery Depends on High-quality Tools
Robotic-assisted surgery has changed what is possible in the operating room. These systems give surgeons better control, steadier hands, and access to areas of the body that are hard to reach. The technology is impressive and deserves the attention it gets. But even the best robot-assisted surgery system depends on the tools that do the…
Read MoreWhy Medical Device Startups Thrive with Medical Murray
At Medical Murray, we don’t just “work with” startups, we love startups. We’ve built our name on transforming early-stage concepts into breakthrough medical devices, particularly with complex catheters and transcatheter implants. Our entrepreneurial spirit fuels a close-knit, collaborative culture where new ideas flourish, and we move quickly from concepts to prototypes, and eventually to pilot…
Read MoreISO 10555 Testing: What Catheter Developers Need to Know
If you are working on an intravascular catheter, there is one standard you need to be familiar with: ISO 10555. This international standard sets the bar for testing catheter performance and plays a key role in design verification and regulatory approval. At Medical Murray, we design and manufacture advanced catheters every day. Our testing capabilities…
Read More